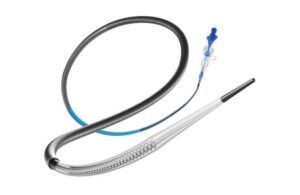
Perfuze wins FDA nod for access catheter
Perfuze announced today that it received FDA 510(k) clearance for its Zipline access catheter and secured significant funding.

Perfuze announced today that it received FDA 510(k) clearance for its Zipline access catheter and secured significant funding.
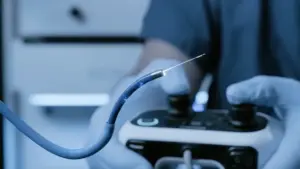
Using Nvidia and GE Healthcare technology, the robotic surgery system is intended to help doctors detect lung cancer earlier.

HERZLIYA, Israel and WILMINGTON, Del., March 13, 2025 /PRNewswire/ — Valcare Medical Inc., a leading innovator in transcatheter-based mitral solutions, today announced the U.S. Food and Drug Administration (FDA) has approved the AMEND™ Trans-Septal System for investigational device exemption (IDE) application to commence an Early Feasibility Study (EFS).

Johnson & Johnson MedTech (NYSE: JNJ)+
announced today that the FDA cleared its Monarch Quest technology for robotic-assisted bronchoscopy.

SALT LAKE CITY, March 12, 2025 /PRNewswire/ — For the thousands of patients in the United States requiring treatment for bone loss or defects caused by trauma or infections, Elute, Inc., a clinical stage company and emerging leader with a groundbreaking controlled and extended drug delivery platform, proudly announces U.S. Food and Drug Administration (FDA) approval of BonVie+™, a novel bone void filler implant.

Sooma Medical announced today that it received FDA investigational device exemption (IDE) for its transcranial direct current stimulation (tDCS) device.
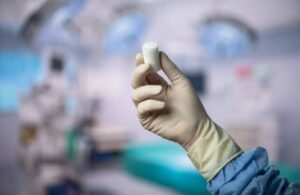
Miach Orthopaedics announced today that the FDA cleared an expanded indication for the company’s Bear implant for ACL tears.

Lungpacer Medical announced today that it received FDA investigational device exemption (IDE) to begin a trial for its AeroNova system.

BREA, Calif., March 10, 2025 /PRNewswire/ — Beckman Coulter Diagnostics, a clinical diagnostics leader, today announced that the new DxC 500i Clinical Analyzer, an integrated clinical chemistry and immunoassay analyzer, received 510(k) clearance from the U.S. Food and Drug Administration. The DxC 500i combines advanced technology with an intuitive user interface, ensuring that laboratories of all sizes can meet the growing demands of modern healthcare. With throughput of up to 800 clinical chemistry tests per hour and 100 immunoassay tests per hour, this analyzer delivers precise and reliable results critical for timely clinical decision-making.

Zimmer Biomet (NYSE: ZBH) announced today that it received FDA 510(k) clearance for its Persona Revision SoluTion Femur.