
FDA clears Nalu peripheral nerve stim tech for whole-body MRI
Nalu Medical announced today that the FDA cleared expanded labeling of its peripheral nerve system to include whole-body MRI use.

Nalu Medical announced today that the FDA cleared expanded labeling of its peripheral nerve system to include whole-body MRI use.

SUNNYVALE, Calif., Jan. 21, 2025 /PRNewswire/ — Inflammatix, a pioneering host response diagnostics company, today announced that the U.S. Food and Drug Administration (FDA) has granted marketing authorization for the TriVerity™ Test System (TriVerity), a first-in-class molecular test for patients with suspected acute infection or sepsis. Using precise measurements of a patient’s immune response, TriVerity combines highly accurate bacterial-viral infection scoring with an all-cause illness severity risk evaluation, giving clinicians a rapid and holistic snapshot of a patient’s status.

HOUSTON, Jan. 21, 2025 /PRNewswire/ — IntelliGenome, an innovative molecular diagnostic solutions provider, is pleased to announce that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation to its CRISPR-Tuberculosis (TB) Blood Test. This test is the first qualitative real-time polymerase chain reaction (PCR) assay to combine CRISPR technology, designed to detect Mycobacterium tuberculosis (Mtb) cell-free DNA in human serum and EDTA plasma.

SARATOGA, Calif., Jan. 21, 2025 /PRNewswire/ — CapsoVision, Inc., a leader in innovative endoscopic capsule technology, today announced that its award-winning product, the CapsoCam Plus® capsule endoscopy system, has received U.S. Food & Drug Administration (FDA) clearance for remote ingestion.

CergenX announced today that the FDA granted breakthrough device designation for its Wave device for neonatal brain monitoring.

ENGLEWOOD CLIFFS, N.J., Jan. 13, 2025 /PRNewswire/ — BRAIN.Q, is excited to announce the CE Mark for its BQ 2.0 system, designed to reduce disability following ischemic stroke, the number one cause of disability worldwide.
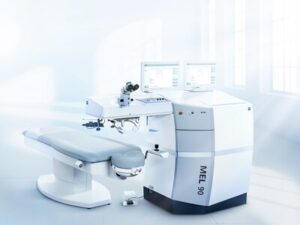
DUBLIN, Calif. and JENA, Germany, Jan. 13, 2025 /PRNewswire/ — ZEISS Medical Technology announced today that the MEL® 90 received approval from the U.S. Food and Drug Administration (FDA), giving the excimer laser technology simultaneous approval for all three major indications, including myopia, hyperopia and mixed astigmatism (a condition where both hyperopic and myopic correction is required).
WOBURN, Mass., Jan. 13, 2025 /PRNewswire/ — Spear Bio Inc., a biotechnology company pioneering next-generation ultra-sensitive immunoassays, today announced that its pTau 217 blood test has been granted Breakthrough Device Designation by the U.S. Food and Drug Administration (FDA). This recognizes the test’s potential to address a critical unmet need for the millions of Americans living with Alzheimer’s disease and not yet diagnosed.

CapsoVision announced today that the FDA cleared its CapsoCam Plus system for use in pediatric patients aged two and above.

TUCSON, Ariz., Jan. 9, 2025 /PRNewswire/ — Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today that its whole slide imaging system, Roche Digital Pathology Dx, has received an additional 510(k) clearance from the United States Food and Drug Administration (FDA).