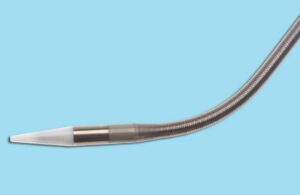
Alleviant Medical wins FDA IDE, breakthrough nods for no-implant heart failure treatment
Alleviant Medical announced today that it received FDA investigational device exemption (IDE) and breakthrough device designation for its atrial shunt.

Alleviant Medical announced today that it received FDA investigational device exemption (IDE) and breakthrough device designation for its atrial shunt.

AccurKardia announced today that it received FDA breakthrough device designation for its ECG-based, AI-powered hyperkalemia detection software.

Medtronic (NYSE: MDT)+
today announced CE mark approval for its Harmony transcatheter pulmonary valve (TPV) system
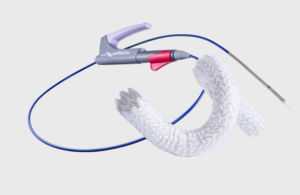
Merit Medical Systems (Nasdaq:MMSI) announced today that it received FDA premarket approval for its Wrapsody cell-impermeable endoprosthesis.

Onward Medical announced today that it received FDA de novo clearance for its ARC-EX spinal cord stimulation (SCS) system.
Roivios Initiates Landmark Study to Demonstrate Safety and Efficacy of an Innovative Renal Assist Device for Cardiac Surgery Patients With Kidney Disease
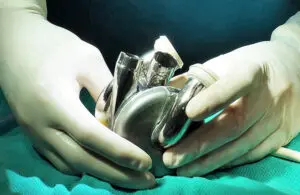
BiVacor announced today that it completed the first phase of an FDA early feasibility study (EFS) for its Total Artificial Heart (TAH).

Smith+Nephew (NYSE: SNN)+
announced that it received FDA clearance for its stemless anatomic total shoulder for the Aetos system.

DÜSSELDORF, Germany, Dec. 16, 2024 /PRNewswire/ — Gerresheimer, an innovative system and solution provider and a global partner for the pharma, biotech and cosmetics industries, announces that the US Food and Drug Administration (FDA) granted SQ Innovation Tentative Approval for Lasix ONYU for the home treatment of fluid overload in congestive heart failure.

The Osseofit devices are intended to match patients’ shoulder bone anatomy and preserve healthy bone in total shoulder replacement procedures.