
FDA clears second set of changes to iRhythm Zio AT to resolve warning letter
iRhythm Technologies (Nasdaq:IRTC) announced that the FDA granted 510(k) clearance for Zio AT design modifications and labeling updates.

iRhythm Technologies (Nasdaq:IRTC) announced that the FDA granted 510(k) clearance for Zio AT design modifications and labeling updates.

Exactech announced today that it received FDA 510(k) clearance for its Truliant porous tibial tray, a 3D tibial knee implant.

Abbott (NYSE:ABT) today announced a significant regulatory milestone that could make common medical procedures easier for people with diabetes.

CytexOrtho today said it has approval from the FDA to start a Phase 1 clinical trial evaluating the safety and efficacy of its absorbable hip implant in human patients.

Paragonix Technologies this week announced it received FDA 510(k) clearance for its KidneyVault donor kidney preservation system.

Distalmotion announced today that it received FDA de novo approval for its Dexter surgical robot for adult inguinal hernia repair.
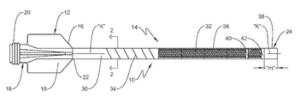
Vantis Vascular announced today that it received FDA 510(k) clearance for its CrossFast integrated microcatheter guide extension system.
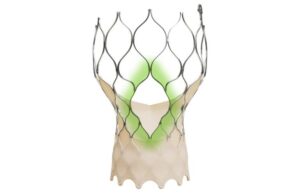
Medtronic (NYSE:MDT) announced today that it received CE mark approval for its Evolut FX+ transcatheter aortic valve implantation (TAVI) system.

Medtronic (NYSE:MDT) today said it received FDA approval for its Affera mapping and ablation system with the Sphere-9 catheter.

The agency cleared one of two 510(k) submissions iRhythm filed for the heart monitor after receiving a warning letter from the agency last year.