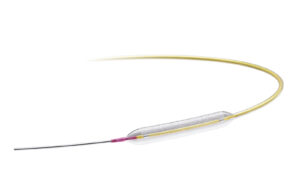
FDA approves IDE for Medtronic Prevail drug-coated balloon
Medtronic (NYSE: MDT)+
announced today that it received FDA investigational device exemption (IDE) for its Prevail drug-coated balloon (DCB).

Medtronic (NYSE: MDT)+
announced today that it received FDA investigational device exemption (IDE) for its Prevail drug-coated balloon (DCB).

A potentially cost-saving UK digital health innovation that is awaiting NHS England funding, more than two years after NICE recommended its national adoption in the NHS, will soon be made available to patients in the US after it secured FDA clearance.

Gentuity announced today that it received FDA 510(k) clearance for its HF-OCT imaging system with the Vis-Rx micro-imaging catheter.

Peytant Solutions announced today that the FDA granted marketing authorization for its AMStent tracheobronchial covered stent system.

Siemens Healthineers says a new FDA clearance will advance the care of people at risk of heart attacks in the United States.
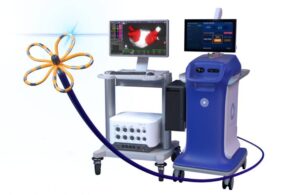
Boston Scientific (NYSE: BSX)+
announced today that it received FDA approval for its navigation-enabled Farawave Nav ablation catheter and clearance for new Faraview software.

Novocure announced on Tuesday that the FDA treated its Optune Lua wearable device for treating metastatic non-small cell lung cancer.
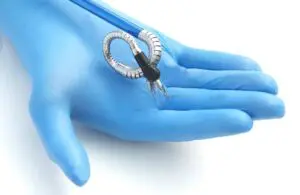
Momentis Surgical (formerly Memic) announced today that it received FDA 510(k) clearance for its Anovo robotic surgical platform.
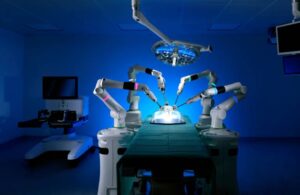
CMR Surgical announced today that it received FDA de novo clearance to market its Versius surgical robot platform.

Medtronic (NYSE: MDT)+ announced today that the FDA approved an early feasibility study to evaluate its Affera system for treating VT.