FDA clears tissue scanner from SamanTree Medical
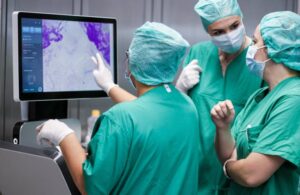
SamanTree Medical announced today that it received FDA 510(k) clearance for its Histolog scanner for tissue imaging.

SamanTree Medical announced today that it received FDA 510(k) clearance for its Histolog scanner for tissue imaging.

SILVER SPRING, Md., Sept. 12, 2024 /PRNewswire/ — Today, the U.S. Food and Drug Administration authorized the first over-the-counter (OTC) hearing aid software device, Hearing Aid Feature, intended to be used with compatible versions of the Apple AirPods Pro headphones. Once installed and customized to the user’s hearing needs, the Hearing Aid Feature enables compatible versions of the AirPods Pro to serve as an OTC hearing aid, intended to amplify sounds for individuals 18 years or older with perceived mild to moderate hearing impairment.

Hinge Health announced that it launched its Enso 3 FDA-cleared wireless device for reducing musculoskeletal pain.

BETHLEHEM, Pa., Sept. 10, 2024 /PRNewswire/ — Tyber Medical LLC, a leading orthopedic device manufacturer specializing in private label implants for the extremity, trauma, and spine markets, is proud to announce that its PEEK ToeGrip Hammer Toe implant family has received Medical Device Regulation (MDR) certification from TÜV Rheinland. This prestigious certification marks the first time a hammer toe implant has achieved both FDA 510(k) clearance and MDR certification, setting a new standard for orthopedic implants.
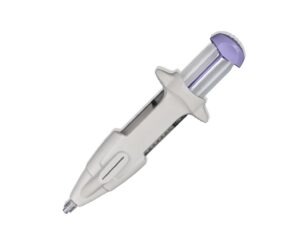
Femasys today announced it received FDA 510(k) clearance for its FemChec contrast-generating device that checks fallopian tubal status.
DeepWell Digital Therapeutics (DTx) has won FDA 510(k) clearance of technology to be used in immersive media like video games for stress reduction and as an adjunctive treatment for high blood pressure.

CENTER VALLEY, Pa., Sept. 5, 2024 /PRNewswire/ — Odin Medical Ltd., an Olympus Corporation company, has received U.S. Food and Drug Administration (FDA) 510(k) clearance for the first cloud-based Artificial Intelligence (AI) technology designed to assist gastroenterologists in detecting suspected colorectal polyps during colonoscopy procedures, the CADDIE™ computer-aided detection (CADe) device.
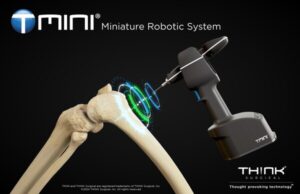
FREMONT, Calif., Sept. 5, 2024 /PRNewswire/ — THINK Surgical, Inc., an innovator in the field of orthopedic surgical robots, today announced that its TMINI® Miniature Robotic System has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for use with the Persona® The Personalized Knee System® from Zimmer Biomet.

The new MRI labelling extends the active scan time for Medtronic DBS systems.

SAN FRANCISCO, Sept. 4, 2024 /PRNewswire/ — Big Health, a leading developer of digital mental health treatments, has been granted clearance by the U.S. Food and Drug Administration (FDA) for its digital therapeutic, DaylightRx. DaylightRx is a prescription device delivering Cognitive Behavioral Therapy and can be made available on the order of a licensed healthcare provider. DaylightRx is a digital therapeutic intended to treat generalized anxiety disorder (GAD) by improving a patient’s GAD symptoms as an adjunct to usual care in patients aged 22 years and older.