
CereVasc wins FDA breakthrough nod for eShunt system
CereVasc announced today that it received FDA breakthrough device designation for its eShunt system for treating normal pressure hydrocephalus (NPH).

CereVasc announced today that it received FDA breakthrough device designation for its eShunt system for treating normal pressure hydrocephalus (NPH).

Neuros Medical announced today that it received FDA approval for its Altius direct electrical nerve stimulation system.
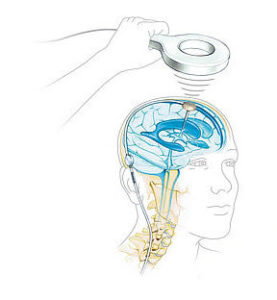
CENTER VALLEY, Pa., Aug. 27, 2024 /PRNewswire/ — Aesculap, Inc. (Aesculap), in partnership with Christoph Miethke GmbH & Co. KG (MIETHKE), announced today that the U.S. Food and Drug Administration (FDA) granted Breakthrough Device Designation for the M.scio® System. This unique, non-invasive, telemetric pressure measurement system is designed to provide continuous access to long-term, intracranial pressure (ICP) monitoring of cerebrospinal fluid (CSF) for the management of hydrocephalus via a permanent, fully implantable sensor.
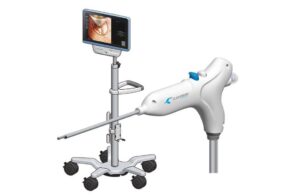
Clearmind Biomedical announced today that it received FDA 510(k) clearance for its Neuroblade neuroendoscopy system.
SUNRISE, Fla., Aug. 26, 2024 /PRNewswire/ — Obvius Robotics, a medical device company developing an innovative technology platform for democratizing vascular access, today announced the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation for its CERTA Access System for central venous catheterization (CVC).
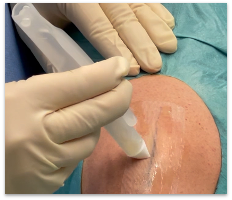
AKRON, Ohio, Aug. 23, 2024 /PRNewswire/ — Resivant Medical announced today it has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its first two products, Cutiva™ Topical Skin Adhesive, and Cutiva PLUS™ Skin Closure System, which combines an adhesive mesh patch with high-viscosity Cutiva™ liquid adhesive. This achievement represents a significant advancement in medical tissue adhesive and wound closure technology and marks the first major adhesive advancement in over 25 years.
SurGenTec announced today that it received FDA 510(k) clearance for its proprietary B-MAN bone marrow aspirate kit.

RA’ANANA, Israel, Aug. 22, 2024 /PRNewswire/ — Inspira Technologies OXY B.H.N. Ltd. (Nasdaq: IINN) (Nasdaq: IINNW) (“Inspira” or the “Company”), a leader in life-support technology innovation, today announces the U.S. Food and Drug Administration (“FDA”) listing of its medical cart (INSPIRA™ CART) which has been custom designed for use with the INSPIRA™ ART100 system.
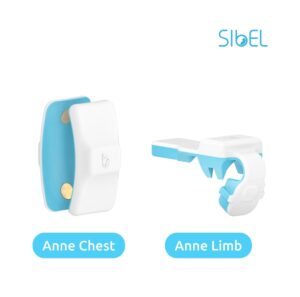
CHICAGO, Aug. 22, 2024 /PRNewswire/ — Sibel Health announces two additional FDA-clearances enabling its advanced wearable sensors to operate with compatible third party software applications. In order to democratize vital signs globally, Sibel Health is committed to enabling widespread access to its advanced wearable sensors.
SAN FRANCISCO and MILWAUKEE, Aug. 21, 2024 /PRNewswire/ — Cytovale®, a commercial-stage medical diagnostics company focused on advancing early detection technologies to diagnose fast-moving and immune-mediated diseases, today announced that the Froedtert & the Medical College of Wisconsin health network is now screening patients with IntelliSep® in a pilot at its flagship hospital, Froedtert Hospital in Milwaukee.