
SoClean wins FDA de novo nod for CPAP, ventilation hose cleaner
SoClean this week announced it was granted FDA de novo clearance for its at-home CPAP mask and ventilation hose cleaner SoClean 3+.

SoClean this week announced it was granted FDA de novo clearance for its at-home CPAP mask and ventilation hose cleaner SoClean 3+.

Si-Bone this week announced it received FDA 510(k) clearance and FDA breakthrough device designation for its iFuse Torq TNT Implant System.

Abbott (NYSE: ABT)+
announced today that the FDA approved a label change for its HeartMate 3 left ventricular assist device (LVAD).
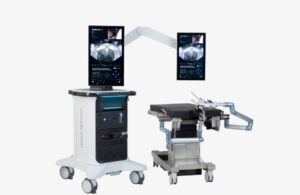
Procept BioRobotics (Nasdaq:PRCT) announced today that it received FDA 510(k) clearance for its next-generation Hydros robotic surgery system.

AlgoDx, a leading medical AI company with EU and UK-approved ML-based software for sepsis prediction in commercial use, has announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its sepsis detection software using a proprietary algorithm, NAVOY CDS. This milestone marks a major step forward in AlgoDx’s expansion into the U.S. market.
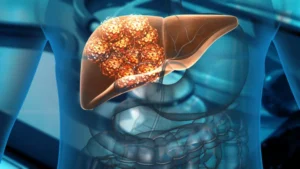
Following a nearly decade-long effort, Delcath Systems finally won the FDA’s greenlight for its Hepzato Kit for the liver-directed treatment of adult patients with metastatic uveal melanoma.
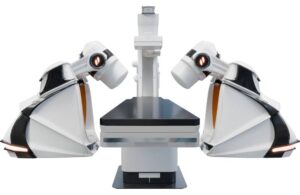
Stereotaxis (NYSE:STXS) announced that it obtained CE mark in Europe and submitted an FDA 510(k) application for its next-generation surgical robot.
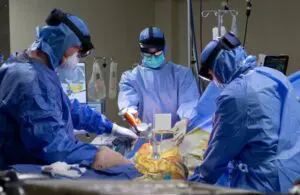
Surgical Planning Associates announced today that it received FDA 510(k) clearance for its HipInsight 2.0 mixed-reality guidance system.
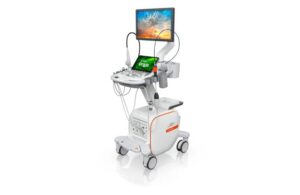
Siemens Healthineers announced today that it received FDA clearance for its Acuson Origin ultrasound system and AcuNav Lumos 4D ICE catheter.
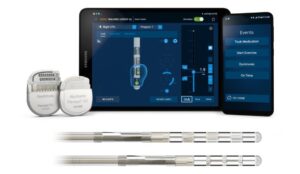
Medtronic says the approval provides more options to meet patients’ personalised needs, including those with Parkinson’s disease.