
Implantable device could save diabetes patients from dangerously low blood sugar
The new implant carries a reservoir of glucagon that can be stored under the skin and deployed during an emergency — with no injections needed.

The new implant carries a reservoir of glucagon that can be stored under the skin and deployed during an emergency — with no injections needed.
Georgia Tech engineers have created a pill that could effectively deliver insulin and other injectable drugs, making medicines for chronic illnesses easier for patients to take, less invasive, and potentially less expensive.

Lifecare said on social media that it completed a significant milestone in the development of its continuous glucose monitor (CGM) implant.

In a clinical trial led by University of Toronto researchers, an allogeneic stem cell–derived islet therapy (zimislecel) restored insulin production and ended severe hypoglycemia in adults with type 1 diabetes within a year of treatment.

Sequel Med Tech announced at the American Diabetes Association’s 85th Scientific Sessions in Chicago that it plans to launch its twiist system in July.

New Clinical Data Shows Significant Delay in Disease Progression and Cognitive Decline in Alzheimer’s Patients Using Cognito’s Wearable Neurotech
Researchers from the Keck School of Medicine of USC have developed a blood test that can identify early signs of Alzheimer’s disease by measuring proteins linked to the condition. The new test, known as Penta-Plex Alzheimer’s Disease Capture Sandwich Immunoassay (5ADCSI), detects five biomarkers simultaneously, which is more than existing blood tests and runs on equipment commonly used in many laboratories.

Go-Pen ApS announced recently that it received FDA 510(k) clearance for its Go-Pen cost-effective, user-filled insulin pen.
A subsidiary of Tandem Diabetes Care (Nasdaq:TNDM) recently picked up FDA clearance for infusion technology to support insulin delivery
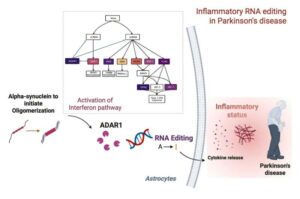
The research team, led by Professor Minee L. Choi from the Department of Brain and Cognitive Sciences, in collaboration with University College London (UCL) and the Francis Crick Institute, discovered that the RNA-editing enzyme ADAR1 plays an important role in controlling immune responses in astrocytes, glial cells that trigger protective reactions in the brain, and demonstrated that this mechanism is critically involved in the progression of Parkinson’s disease.