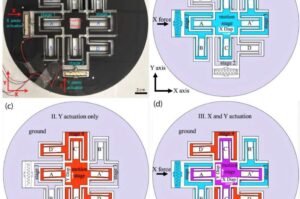
Vibration-powered chip could revolutionize assisted reproductive technology
In the quest to address infertility, Cornell researchers have developed a groundbreaking device that could simplify and automate oocyte cumulus removal, a critical step in assisted reproductive technologies.