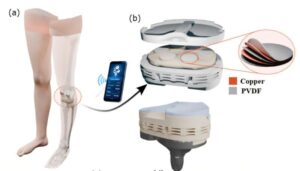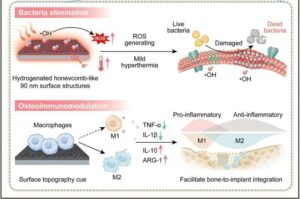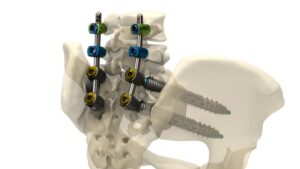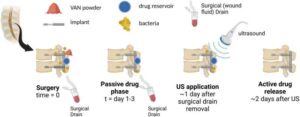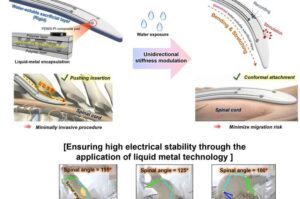
Spinal cord stimulator stays rigid for surgery, then softens inside the body
What if chronic diseases, which are difficult to treat with medicine alone, could be managed with electricity? As “neuromodulation”—a technology that restores bodily balance by sending signals directly to nerves—gains attention, a Korean research team has brought this possibility one step closer to reality.