Renovos secures FDA breakthrough status for bone graft alternative
When injected, Renovite acts as a scaffold for cells and helps localize and retain molecules that stimulate healing

When injected, Renovite acts as a scaffold for cells and helps localize and retain molecules that stimulate healing
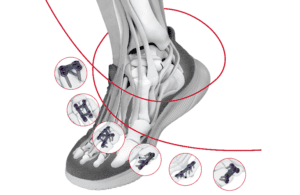
DePuy Synthes, the orthopedic device business of Johnson & Johnson (NYSE: JNJ)+
, has won FDA 510(k) clearance for its TriLeap lower extremity anatomic plating system.

ZKR Orthopedics, Inc., a clinical stage medical device company, today announced that its LIFT implant technology has been granted Breakthrough Device Designation by the U.S. Food and Drug Administration (FDA).
Moximed announced today that doctors completed the first commercial cases for its Misha knee system, an implantable shock absorber (ISA).