
MONARCH Platform for Bronchoscopy Receives Regulatory License for China
The MONARCH™ Platform is the first minimally invasive, robotic-assisted technology approved for bronchoscopy in a country with significant burden of lung cancer.

The MONARCH™ Platform is the first minimally invasive, robotic-assisted technology approved for bronchoscopy in a country with significant burden of lung cancer.
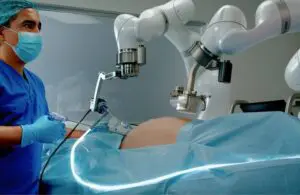
Levita Magnetics today announced the world’s first commercial use for its MARS magnetic-assisted robotic surgery platform.
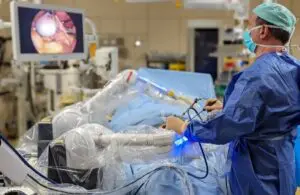
Moon Surgical today announced the successful completion of the first 10 laparoscopic surgeries with its Maestro system in France.

Interventional Systems announced that the FDA granted a new 510(k) clearance for its Micromate surgical robot system.

The startup affiliated with Johns Hopkins University said it is opening a bridge round for its series B financing as it targets more applications for the robotic surgery assistant.
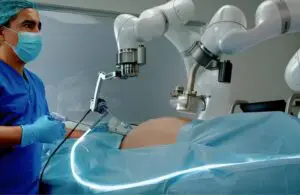
Mountain View, California-based Levita designed its MARS minimally invasive surgical platform for the high-volume abdominal surgery market. It combines magnets and machines to reduce the number of incisions, enabling complete control during laparoscopic procedures.

A tiny robot which can travel deep into the lungs to detect and treat the first signs of cancer has been developed by researchers at the University of Leeds.