Neocis unveils next-gen AI-powered dental robot
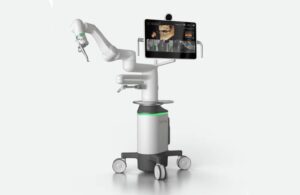
Neocis announced today that it launched Yomi S, its next-generation robotic platform for dental implant surgery.

Neocis announced today that it launched Yomi S, its next-generation robotic platform for dental implant surgery.

Competing with companies including Stryker and J&J, Zimmer has moved to strengthen its position by developing an updated robot.
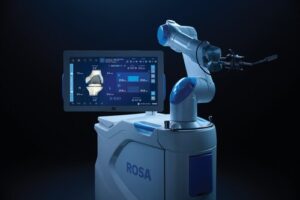
WARSAW, Ind., Nov. 14, 2025 /PRNewswire/ — Zimmer Biomet Holdings, Inc. (NYSE and SIX: ZBH), a global medical technology leader, today announced U.S. Food and Drug Administration (FDA) 510(k) clearance of ROSA® Knee with OptimiZe™, an enhanced version of its ROSA® Knee System that offers a more customized experience for surgeons to help deliver accurate and reproducible outcomes1 in robotic-assisted total knee replacement surgery.
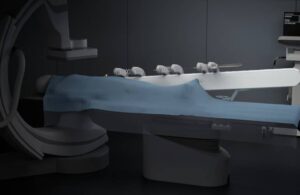
Sentante today announced the completion of a first-of-its-kind remote stroke procedure in Scotland using its robotic platform.
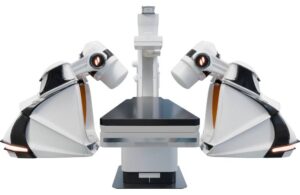
Stereotaxis (NYSE:STXS) announced today that it received FDA 510(k) clearance for its next-generation GenesisX surgical robot.
Medical Microinstruments (MMI) announced today that it received FDA investigational device exemption (IDE) to study its microsurgery platform

Wandercraft announced today that the FDA expanded the indications for its flagship Atalante X rehabilitation device.
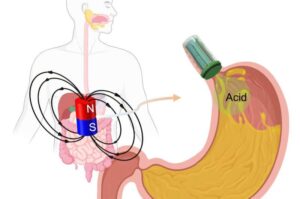
A team of researchers led by Xiaoguang Dong, assistant professor of mechanical and biomedical engineering at Vanderbilt University, have developed a magnetic robotic valve to provide minimally invasive intervention for gastroesophageal reflux disease and possibly other organ system disorders.

Edge Medical announced on LinkedIn today that it received CE mark for MSP2000, its robotic-assisted “super system.”
https://www.medtechdive.com/news/stereotaxis-teams-with-cardiofocus-to-develop-robotic-pfa-system/802799/