
LEM Surgical wins FDA clearance for hard tissue surgical robot
LEM Surgical announced that it received FDA 510(k) clearance for its Dynamis robotic surgical system for hard tissue surgery.

LEM Surgical announced that it received FDA 510(k) clearance for its Dynamis robotic surgical system for hard tissue surgery.

Discover the groundbreaking Betavolt atomic battery, redefining energy with a 50-year charge cycle.
FREMONT, Calif., April 16, 2025 /PRNewswire/ — THINK Surgical, Inc., an innovator in the field of orthopedic surgical robots, today announced the successful first use of Maxx Orthopedics’ Freedom Total Knee implant utilizing the TMINI® Miniature Robotic System by Dr. David V. Cashen, a Joint Replacement specialist at Coastal Orthopedics Surgery Center in Bradenton, Florida.

Intuitive (Nasdaq: ISRG) announced today that it received FDA clearance for its fully wristed SP SureForm 45 stapler.

Zeiss Medical Technology announced today that it received FDA 510(k) clearance for the Intrabeam 700 platform.
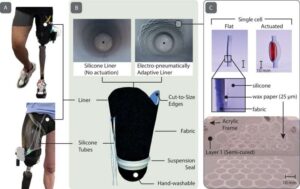
The latest advance in wearable robotic technology promises to solve a 200-year-old problem by revolutionizing the fit of prosthetic limbs, transforming the lives of millions of amputees worldwide.
Monogram Technologies secured 510(k) clearance for its robotic knee replacement system

Moon Surgical announced today that it received FDA clearance for ScoPilot, a Nvidia-enabled platform for its Maestro surgical robot.

Lifeward (Nasdaq:LFWD) announced today that it received FDA 510(k) clearance for its latest-generation personal exoskeleton device, ReWalk 7.

Spineart and eCential Robotics today announced the receipt of FDA 510(k) clearance for the use of an application for robotic navigation.