
FDA grants 510(k) clearance to TaeWoong Medical for Spaxus EUS stent
TaeWoong Medical, the fully covered, self-expanding metallic device, is intended to aid EUS drainage procedures.

TaeWoong Medical, the fully covered, self-expanding metallic device, is intended to aid EUS drainage procedures.

https://medtechspectrum.com/news/40/25517/unm-scientist-studies-where-brain-cancer-cells-hide-and-how-to-fight-them.html
Scientists have developed a way to turn the body’s own immune cells into cancer-fighting agents—without removing them from the body—by using red blood cells to deliver genetic instructions.

A first-in-human clinical trial led by an international team of researchers and published in the New England Journal of Medicine found that setidegrasib, an investigational targeted therapy drug designed to eliminate a key cancer-driving protein called KRAS G12D, shows encouraging early activity in patients with advanced lung and pancreatic cancers.
Founded by Jake Donoghue PhD ’19 and former MIT researcher Jarrett Revels, the company is creating an AI-driven platform to help diagnose and treat disease.

Philips (NYSE:PHG) announced that it received FDA 510(k) clearance for its Smart Fit TorsoCardiac magnetic resonance (MR) 1.5T coil.
Agitated Solutions announced that it received CE mark approval for its novel Orbis microbubble generator device.

Zeta Surgical announced today that the FDA granted 510(k) clearance for its Zeta Navigation System and its navigated Stylet and Bolt instruments.
The U.S. Food and Drug Administration has approved Merck’s Idvynso (doravirine/islatravir), a new, once-daily, two-drug single tablet for the treatment of HIV-1 infection in adults to replace the current antiretroviral regimen in those who are virologically suppressed (HIV-1 RNA <50 copies per mL).
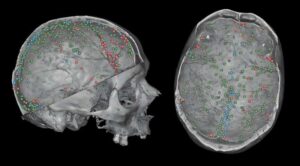
A study led by Rafael Gallareto-Sande, a predoctoral researcher at the Centro Nacional de Investigación sobre la Evolución Humana (CENIEH), provides new insights into a network of tiny blood vessels within the cranial bones that play a key role in brain protection.