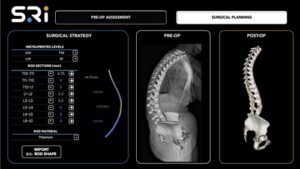
Spinal Resources, Inc. Awarded Second U.S. Patent for Multi-Diameter Spinal Fixation Rod
FORT LAUDERDALE, Fla., May 7, 2026 /PRNewswire/ — Spinal Resources, Inc., a developer of advanced spinal stabilization technologies, today announced the issuance of U.S. Patent No. 12,611,231, “Spinal Rod and Systems Thereof”.