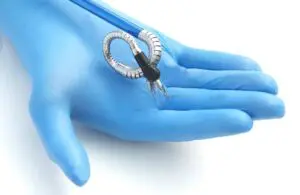
Momentis Surgical wins FDA clearance for its surgical robot
Momentis Surgical (formerly Memic) announced today that it received FDA 510(k) clearance for its Anovo robotic surgical platform.

Momentis Surgical (formerly Memic) announced today that it received FDA 510(k) clearance for its Anovo robotic surgical platform.
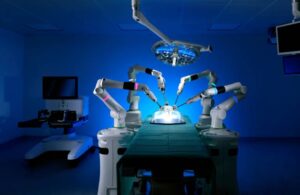
CMR Surgical announced today that it received FDA de novo clearance to market its Versius surgical robot platform.

Paragonix today announced the first pediatric use of its BAROguard Donor Lung Preservation System in a lung transplant case.
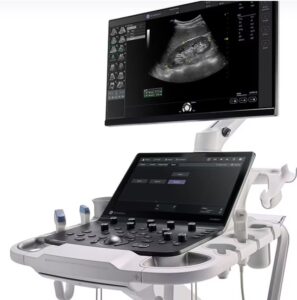
GE HealthCare (Nasdaq: GEHC)+
announced that it launched the new Versana Premier, its latest offering from the Versana family of ultrasound systems.
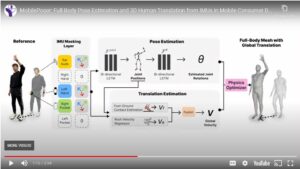
MobilePoser makes motion capture more accessible by using sensors your devices already have

Promising findings by researchers at Baylor College of Medicine and collaborating institutions could lead to the development of a non-invasive stool test and a new therapy for endometriosis, a painful condition that affects nearly 200 million women worldwide. The study appears in the journal Med.

Medtronic (NYSE: MDT)+ announced today that the FDA approved an early feasibility study to evaluate its Affera system for treating VT.
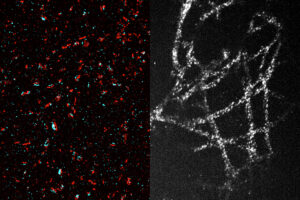
Labs that can’t afford expensive super-resolution microscopes could use a new expansion technique to image nanoscale structures inside cells.

Iota Biosciences, a subsidiary of Astellas Pharma, announced that it received FDA investigational device exemption (IDE) for its bladder implant.

Endostart announced today that it received CE mark approval for the expanded use of its flagship Endorail product.