
Qnovia wins FDA nod for nicotine inhaler as smoking cessation therapy
Qnovia announced that the FDA cleared its investigational new drug (IND) application for the RespiRx smoking cessation inhaler (QN-01).

Qnovia announced that the FDA cleared its investigational new drug (IND) application for the RespiRx smoking cessation inhaler (QN-01).
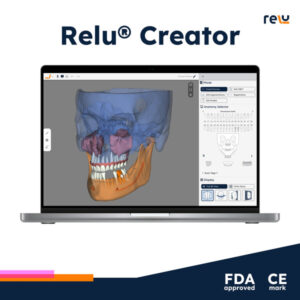
LEUVEN, Belgium, Oct. 1, 2024 /PRNewswire/ — Relu, a pioneer in artificial intelligence (AI) assisted segmentation for dental labs and software companies, proudly announces the dual achievement of 510(k) clearance by the U.S. Food and Drug Administration (FDA) and CE Mark approval by an EU Notified Body. These regulatory milestones authorize the commercial distribution of the Relu® Creator, the cutting-edge dental tool that enables users to create 3D anatomical models from patients in just minutes.

Surmodics (Nasdaq:SRDX) announced today that it received FDA 510(k) clearance for its Pounce XL thrombectomy system.

Called Flyrcado, the radiotracer could help improve cardiac imaging accuracy in patients with a high body mass index and women, the company said.

Ascensia Diabetes Care announced today that it launched the Eversense 365 CGM system from Senseonics (NYSE:SENS) in the U.S.

Stevanato Group announced today that it expanded the capacity of its Vertiva on-body device for subcutaneous large-volume drug delivery.

Johnson & Johnson (NYSE: JNJ)+
announced today that it expanded the rollout of its latest Tecnis Odyssey presbyopia-correcting intraocular lenses (PC-IOLs).

Pi-Cardia announced today that the FDA granted clearance for its ShortCut leaflet modification device for TAVR procedures.
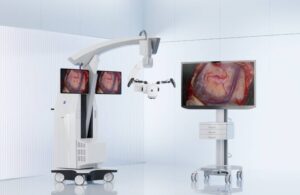
Zeiss Medical Technology announced today that it launched the Kinevo 900 S robotic visualization system for neurosurgery procedures.

ResMed (NYSE: RMD)+
announced today that it launched its new AirTouch N30i continuous positive airway pressure (CPAP) mask.