
Spineart wins FDA clearance for Scarlet AC-Ti anterior cervical cage
Spineart recently announced it received FDA 510(k) clearance for its Scarlet AC-Ti secured anterior cervical cage.

Spineart recently announced it received FDA 510(k) clearance for its Scarlet AC-Ti secured anterior cervical cage.

Terumo Cardiovascular today announced FDA 510(k) clearance of its next-generation CDI OneView monitoring system.

BUENA, N.J., May 29, 2024 /PRNewswire/ — COMAR, a leading provider of innovative medical devices and packaging solutions, is thrilled to announce a significant achievement. After rigorous preparation, they successfully obtained their CE certificate under the latest EU Medical Device Regulations (EU MDR 2017/745).
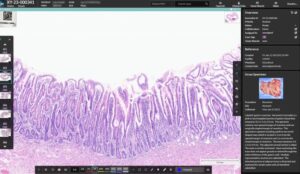
HALO AP Dx brings digital primary diagnosis to anatomic pathology labs in the US.

Canary Medical announced today that it received FDA breakthrough device designation for its Canturio lumbar cartridge.
Medtronic (NYSE: MDT)+
announced today that the FDA granted breakthrough device designation for its Infuse bone graft.

RA’ANANA, Israel, May 28, 2024 /PRNewswire/ — Inspira™ Technologies OXY B.H.N. Ltd. (Nasdaq: IINN) (Nasdaq: IINNW) (the “Company”, “Inspira”, or “Inspira Technologies”), a breakthrough medical technology company, today announced that it has received notification of 510(k) class II clearance from the U.S. Food and Drug Administration (FDA) for its INSPIRA™ ART100, a Cardiopulmonary Bypass System.

Galvanize announced today that the FDA granted 510(k) clearance for its Inumi Flex endoscopic needle with the Aliya system for soft tissue ablation.
Ethicon announced that it began the U.S. launch of its first-of-its-kind Echelon Linear surgical stapler.
New research suggests the HPV vaccine is preventing cancer in men, as well as in women, but fewer boys than girls are getting the shots in the United States.