
MMI receives de novo nod for microsurgery robot
Medical Microinstruments said the system could increase the number of physicians who can perform complicated microsurgical procedures.

Medical Microinstruments said the system could increase the number of physicians who can perform complicated microsurgical procedures.

Neurovalens announced today that it received FDA clearance for its Modius Stress device for treating anxiety and raised $2.65 million.

Orthobond has secured FDA de novo approval for its Ostaguard antibacterial technology that could one day be used for a wide range of medical devices — and beyond medtech.
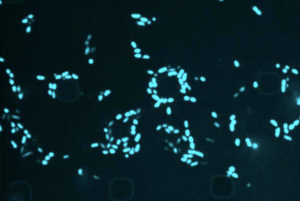
Most antibiotics target metabolically active bacteria, but with artificial intelligence, researchers can efficiently screen compounds that are lethal to dormant microbes.

Siemens Healthineers announced today that it received FDA 510(k) clearance for its MammoMat B.brilliant mammography platform.

New Indication for Treatment of Pulmonary Embolism Enhances Device Utility in Critical Medical Scenarios

This method of use patent award marks a major achievement for the Company and follows another recent patent covering our core technology.
The clearance adds to the list of devices the FDA has authorized this year with AI algorithms to detect health conditions.

The CEO of Prenosis told MedTech Dive the company sees third-party validation as important, with the FDA having clarified that certain decision support tools should be regulated as medical devices.

The low-cost hardware outperforms state-of-the-art versions and could someday enable an affordable, in-home device for health monitoring.