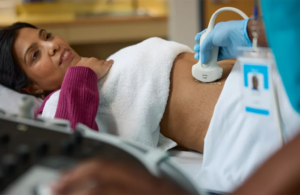
Philips wins FDA clearance for AI-powered ultrasound workflow tech
Philips (NYSE:PHG) announced today that it received FDA clearance for its Elevate Plus advanced imaging and AI and automation capabilities.

Philips (NYSE:PHG) announced today that it received FDA clearance for its Elevate Plus advanced imaging and AI and automation capabilities.
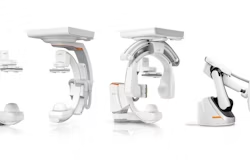
Pulmera announced today that it received FDA 510(k) clearance for its CBeam 3D intraoperative imaging platform.

Edwards Lifesciences (NYSE:EW) said today that it received FDA approval for Triformis Resilia, its surgical tricuspid valve replacement.
Developed by an international team, led by scientists at UCL and at the Allen Institute (Seattle, US), the research forms part of a project investigating Neuropixels probe technology.
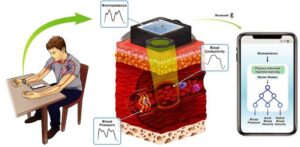
The technology records tiny electrical changes in your wrist using bioimpedance, a measure of how easily electricity flows through blood and tissue. Because blood flow changes with each heartbeat, these electrical signals carry information about the underlying pressure.

The development coincides with several steps by SHINE Technologies to broaden its position in nuclear medicine.
The daily pills nearly doubled survival time, with fewer severe side effects, in a study that randomly assigned the experimental drug or more chemotherapy to 500 patients whose metastatic, or spreading, cancer had quit responding to prior treatment.
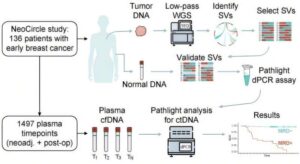
Researchers at Lund University have developed a blood test capable of detecting signs of breast cancer recurrence long before recurrence becomes visible on imaging or causes symptoms.
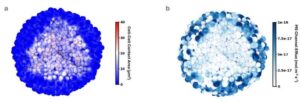
Researchers have solved a long-standing mystery about why physical forces slow cancer growth—and the answer could reshape how the disease is treated. A multidisciplinary team from University of Galway, CÚRAM, the Taighde Éireann-Research Ireland Centre for Medical Devices, and KU Leuven in Belgium built an innovative AI accelerated computational model to test the theory.
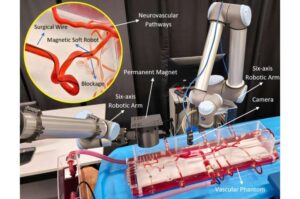
Researchers at Concordia have developed an AI-assisted technique and a robotic platform that may one day help surgeons perform safer, faster and less invasive procedures to treat conditions such as blood clots located deep inside a patient’s neurovascular pathways.