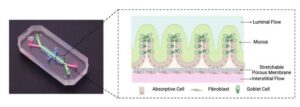
3D-printed trays help human gut organoids self-build nerves and mature twice as fast
Thanks to special 3D-printed scaffolding trays designed by experts at Cincinnati Children’s, researchers can now produce larger versions of functional human gut organoids twice as fast as previous methods—and these organoids grow their own nerve cells.