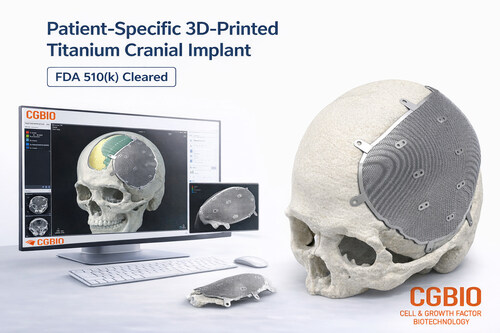
This milestone marks the first case of a Korean company obtaining FDA 510(k) clearance for a patient-specific titanium implant intended for the U.S. market. It highlights CGBIO’s technological capabilities as well as its regulatory expertise in global markets. The achievement is particularly notable in the field of patient-specific cranial and craniofacial reconstruction.
EASYMADE TI is a patient-specific titanium implant designed based on individual CT data and intended for reconstruction of cranial and non-load-bearing craniofacial defects. The implant is manufactured using medical-grade titanium alloy and produced through Laser Powder Bed Fusion (LPBF), an advanced metal 3D printing technology.