
Label-free optical imaging enables automated measurement of human white matter microstructure
In a study published in Biophotonics Discovery, researchers report a new way to examine white matter structure in postmortem human brains.

In a study published in Biophotonics Discovery, researchers report a new way to examine white matter structure in postmortem human brains.
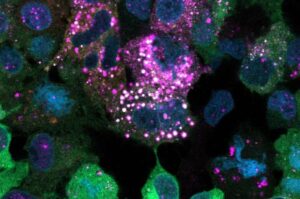
WEHI researchers have discovered a never-before-seen mechanism our bodies use to regulate sugar, in findings that rewrite the fundamental rules of biology and open a new frontier in science.
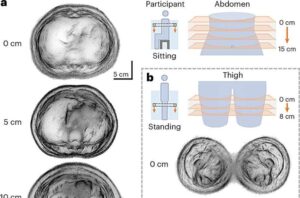
Lihong Wang, Bren Professor of Medical Engineering and Electrical Engineering, and a team of Caltech researchers have developed a system that can perform ultrasound tomography (UST) imaging on whole cross-sections of the body.
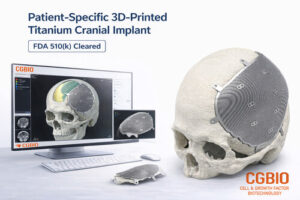
SEOUL, South Korea, April 24, 2026 /PRNewswire/ — CGBIO (CEO: Hyunseung Yu) announced that its patient-specific titanium implant, EASYMADE TI, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) on April 9. The FDA 510(k) pathway is a premarket clearance process that demonstrates substantial equivalence to a legally marketed predicate device, allowing commercialization in the United States.

Medtronic (NYSE:MDT) announced today that it received CE mark approval for its ApexCut surgical blades for ENT procedures.