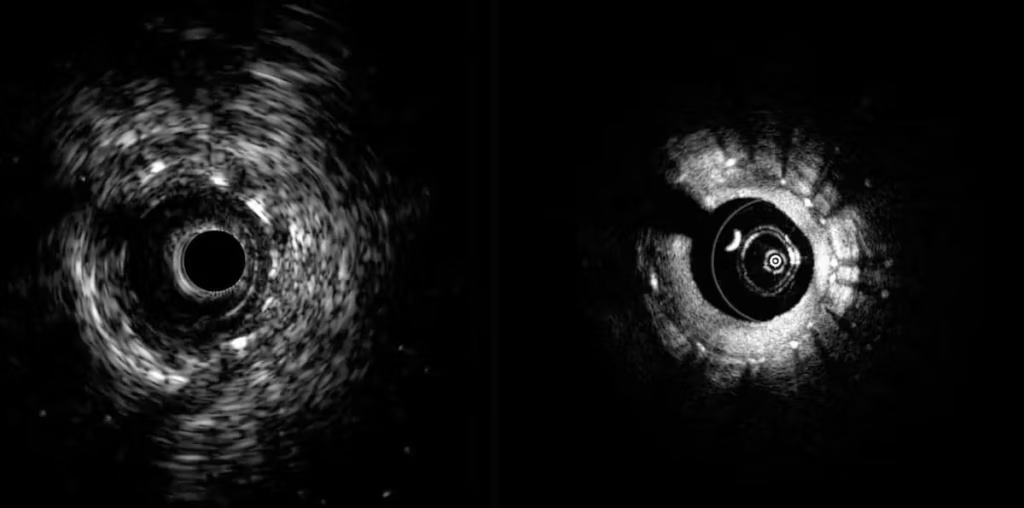
Toronto-based Conavi designed its next-generation system to deliver a more complete assessment of coronary anatomy within a single workflow. The company submitted the platform to the FDA in September 2025.
Conavi’s hybrid system integrates intravascular ultrasound (IVUS) and optical coherence tomography (OCT) in a single platform. Together, these technologies enable physicians to visualize both deep vessel structures and high-resolution surface detail in real time. The company says this supports physician decision-making and streamlines workflows.
According to Conavi, the system’s dual-modality approach can provide a comprehensive view of coronary arteries. This potentially enables more precise diagnosis and treatment of cardiovascular disease.