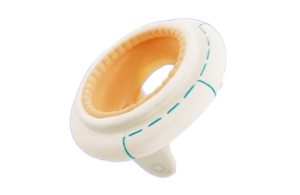
The medtech giant designed the Mosaic Neo mitral bioprosthesis for implantation through sternotomy, minimally invasive cardiac surgery (MICS) and robotic access. Numerous recent procedures across the U.S. performed Mosaic Neo implantations using these methods, the company said. That includes at The Mount Sinai Hospital New York, where Dr. David H. Adams implanted the first Mosaic Neo mitral valves.
Simultaneously, the company reported the first concomitant use of Mosaic Neo with the Penditure left atrial appendage (LAA) exclusion system. Meanwhile, Dr. Vinay Badhwar of the West Virginia University (WVU) Heart and Vascular Institute performed the first robotic implantation.