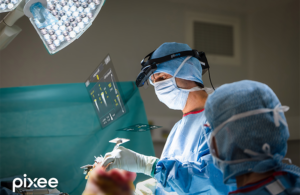
Besançon, France-based Pixee designed its AR-based technology for total knee arthroplasty (TKA) procedures. The company already brought its previous-generation Knee NexSight system to the U.S., announcing the first U.S. procedure in May 2025. It had just unveiled the technology at AAOS in March 2025.
Clearance allows Pixee to bring the latest AR technology to the U.S., following its CE mark and subsequent launch in Europe last month. The company said its focus centers on ambulatory surgery centers (ASCs), with the technology designed to integrate seamlessly into the high-demand environments, streamlining workflows without adding complexity.