
Motif Neurotech wins FDA IDE to evaluate brain implant for treating depression
Motif Neurotech announced today that it received FDA investigational device exemption (IDE) to evaluate its brain stimulation implant.

Motif Neurotech announced today that it received FDA investigational device exemption (IDE) to evaluate its brain stimulation implant.
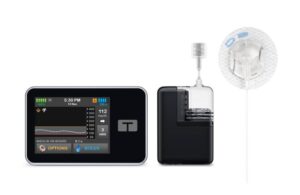
Tandem Diabetes Care (Nasdaq:TNDM) today announced FDA clearance for its automated insulin delivery (AID) technology for use in pregnancy.
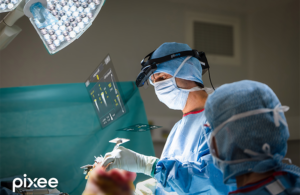
Pixee Medical announced today that it received FDA 510(k) clearance for its Knee+ NexSight augmented reality (AR)-based solution.
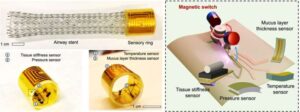
Vanderbilt researchers led by Xiaoguang Dong, assistant professor of mechanical engineering, have developed a novel device that can be used remotely to continuously monitor the airway stents of patients with diseases like lung cancer.

NEW YORK, April 27, 2026 /PRNewswire/ — TytoCare, a virtual care company enabling accessible, high-quality primary care from home, today announced it has received FDA De Novo classification for the first model of Tyto Insights™ for ENT Suite, marking the creation of a new regulatory category for AI-powered ENT image analysis.