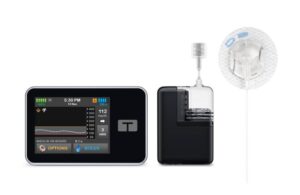
The FDA cleared the company’s Control-IQ AID technology for use in pregnancy complicated by type 1 diabetes mellitus. Control-IQ+ powers both of Tandem’s available t:slim X2 and Mobi AID systems. San Diego-based Tandem says t:slim X2 and Mobi are the first and only commercially available AID systems cleared for use during pregnancy in the U.S.
Control-IQ+ represents the next generation of the company’s advanced hybrid closed-loop insulin delivery algorithm. It accommodates a wider range of insulin requirements and has enhanced extended bolus capabilities up to eight hours when turned on. Control-IQ+ allows for better meal management with delayed glucose rise or prolonged eating. New temporary basal rate adjustments also address short-term glucose needs like exercise, stress or illness.