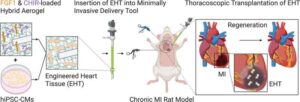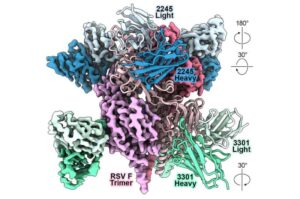
AI can speed antibody design to thwart novel viruses
Artificial intelligence (AI) and “protein language” models can speed the design of monoclonal antibodies that prevent or reduce the severity of potentially life-threatening viral infections, according to a multi-institutional study led by researchers at Vanderbilt University Medical Center.