MedTech News
.................... by Andrew Celentano
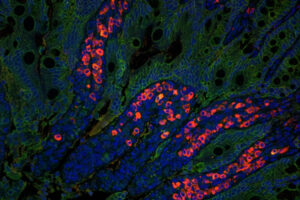
A new vaccine adjuvant could make it easier to eradicate polio
The adjuvant can help the injectable polio vaccine induce a strong immune response in the GI tract, which is considered critical to eradicating the virus.

Real-time brain monitor detects infections earlier
The result is NeuroSense, a monitoring system that connects to drainage lines to detect biomarkers of infection, including changes in glucose, lactate and pH, as well as flow rate, as brain fluid moves through them.

AI helps accelerate breast cancer diagnosis for high-risk women
Researchers used an open-source AI model called Mirai, which was developed by the study’s senior author, UC Berkeley data scientist Adam Yala, Ph.D.
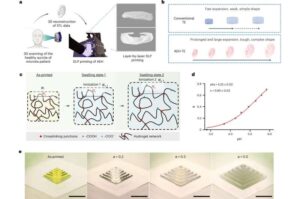
Custom 4D-printed implants offer less painful path to tissue reconstruction
Di Wang, Ph.D., and Y. Shrike Zhang, Ph.D., of the Division of Engineering in the Mass General Brigham Department of Medicine, the lead and senior authors of the study published in Nature Biomedical Engineering, developed a special gel-like material whose expansion speed and final size can be carefully controlled. The paper is titled “4D-printed adaptive hydrogel tissue expanders for ear and breast reconstruction.”
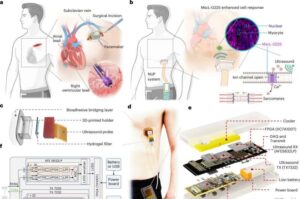
Ultrasound-based pacemaker noninvasively steadies the heart
MIT engineers have developed a noninvasive pacemaker that stimulates the heart using ultrasound. The design could one day provide a surgery-free alternative to traditional cardiac implants.
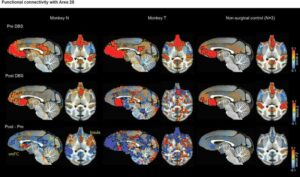
Deep brain stimulation boosts myelination and shifts brain networks linked to depression
Researchers from the Icahn School of Medicine at Mount Sinai have uncovered the first direct evidence that deep brain stimulation (DBS) can remodel white matter pathways in the brain and alter communication across large-scale neural networks, revealing a previously unrecognized mechanism that may explain how the therapy helps patients recover from severe depression.
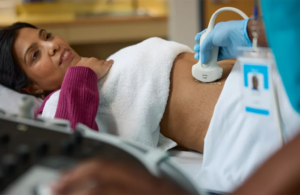
Philips wins FDA clearance for AI-powered ultrasound workflow tech
Philips (NYSE:PHG) announced today that it received FDA clearance for its Elevate Plus advanced imaging and AI and automation capabilities.
Pulmera wins FDA clearance for 3D imaging platform
Pulmera announced today that it received FDA 510(k) clearance for its CBeam 3D intraoperative imaging platform.
