MedTech News
.................... by Andrew Celentano
Blood test with AI spots four dementia-related brain diseases with 92.3% accuracy
Researchers at Washington University School of Medicine in St. Louis have developed an AI-based classifier that distinguishes between four common brain diseases that cause dementia: Alzheimer’s disease, Parkinson’s disease, frontotemporal dementia and dementia with Lewy bodies, as well as healthy brain aging.

SKIA HEAD: AR Surgical Guidance Receives FDA 510(k) Clearance, Eyes U.S. Expansion with Structure Partnership
SEOUL, South Korea, May 29, 2026 /PRNewswire/ — South Korea-based medtech innovator SKIA has announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for its augmented reality (AR) surgical guidance system, SKIA HEAD.

Gene Editing May Help Treat Dangerously High Cholesterol with Just One Infusion
Gene editing may help people born with dangerously high cholesterol lower their levels with just one infusion.

New Tool That Tracks How the Brain Removes Waste Could Offer Clues About Alzheimer’s
How the brain clears its waste could help researchers combat neurodegenerative diseases and age-related cognitive decline.
Coredio’s AI heart failure software secures breakthrough designation
Coredio’s AI software is used to monitor HF parameters via wearables and standard blood pressure cuffs.

MannKind wins FDA approval for inhaled insulin for children with type 1, type 2 diabetes
MannKind (Nasdaq:MNKD) announced today that it received FDA approval for its Afrezza inhaled insulin powder for children.
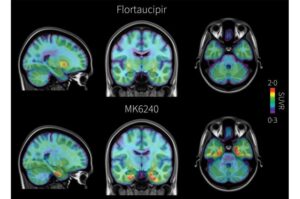
New brain scan detects Alzheimer’s tau earlier than current standard
A new brain imaging test can detect a key hallmark of Alzheimer’s disease before symptoms appear and earlier than the method currently used in clinical practice in the United States and Europe, report University of Pittsburgh School of Medicine researchers in The Lancet.
Lab-grown heart patch boosts pumping power in severe heart failure trial
Researchers at the University Medical Center Göttingen (UMG) and the University Hospital of Schleswig-Holstein (UKSH) have made a significant breakthrough in the treatment of severe heart failure: For the first time, a clinical trial has demonstrated that laboratory-grown heart muscle tissue can improve the pumping function of damaged hearts.
