
Zimmer receives FDA clearance for robotic shoulder system
The clearance poises the orthopedics company to be first to market with robotic-assisted shoulder replacement surgery.

The clearance poises the orthopedics company to be first to market with robotic-assisted shoulder replacement surgery.

For use in children weighing 10 kilograms or more with acute kidney injury (AKI) due to sepsis or a septic condition requiring kidney replacement therapy (KRT).
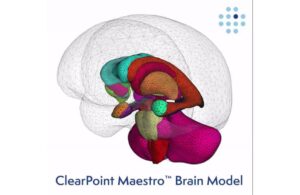
ClearPoint Neuro (Nasdaq:CLPT) announced that the FDA cleared its ClearPoint 2.2 software with integrated Maestro Brain Modeling.

Merit Medical Systems (Nasdaq:MMSI) announced today that it received FDA 510(k) clearance for the Scout MD surgical guidance system

The company’s OTC device, which costs nearly $300, will compete with consumer pulse oximeters that are widely available but lack FDA clearance.

SAN FRANCISCO, Feb. 14, 2024 (GLOBE NEWSWIRE) — ZKR Orthopedics, Inc., a clinical stage medical device company, announced that it has received Investigational Device Exemption (IDE) approval from the United States Food and Drug Administration (FDA) for a multicenter prospective clinical trial. The approved trial will evaluate the Company’s LIFT implant to treat later stage patellofemoral cartilage degeneration in adult patients.
Abbott (NYSE: ABT)+
announced today that an FDA advisory committee ruled that the benefits of its TriClip outweigh the risks in treating tricuspid regurgitation (TR).

Proscia, a leading provider of digital and computational pathology solutions, has received 510(k) clearance from the United States (U.S.) Food and Drug Administration (FDA) for its Concentriq AP-Dx. The digital pathology solution was cleared for the purpose of primary diagnosis.
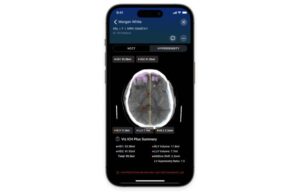
Viz.ai announced that it received FDA 510(k) clearance for its Viz ICH Plus algorithm for providing volume measurements of brain bleeds.

Fresenius Medical Care announced today that it received FDA clearance for its 5008X hemodialysis system.