
Axogen Announces FDA Approval of Biologics License Application for AVANCE
Avance is an acellular nerve scaffold for the treatment of adult and pediatric patients aged 1 month or older with sensory, mixed, and motor peripheral nerve discontinuities

Avance is an acellular nerve scaffold for the treatment of adult and pediatric patients aged 1 month or older with sensory, mixed, and motor peripheral nerve discontinuities
SURREY, BC, Dec. 4, 2025 /PRNewswire/ – mlHealth 360 today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance (K250694) for Scaida BrainCT-ICH, an AI-powered triage solution designed to address critical bottlenecks in acute care radiology.

MIDDLETON, Wis., Dec. 3, 2025 /PRNewswire/ — Natus Medical Incorporated announced the electrographic status epilepticus diagnosis capability of its BrainWatch point-of-care EEG solution, featuring integrated Persyst analysis software which has received 510(k) clearance from the U.S. Food and Drug Administration.

BEIJING, Dec. 3, 2025 /PRNewswire/ — Wingderm® announces that its Renuva 1550nm non-ablative fractional laser system has obtained MDR certification in the European Union, marking a significant regulatory milestone for the device within the EU market.
Medtronic (NYSE: MDT)+
announced today that the FDA cleared its Hugo robotic-assisted surgery system for use in urologic surgical procedures.
The test, which runs on the cobas liat system, is said to deliver results in 15 minutes.
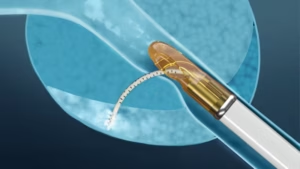
The Vanquish device’s clinical data demonstrated an elimination of MRI visible intermediate risk disease in 91% of patients after a single procedure.
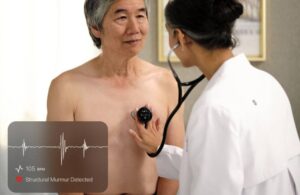
Eko Health announced today that the Centers for Medicare & Medicaid Services (CMS) has finalized national payment for its Sensora platform.

MannKind (Nasdaq:MNKD) announced that the FDA accepted a supplemental New Drug Application (sNDA) for its Furoscix ReadyFlow autoinjector.

The decision is supported by evidence from a clinical study and data from supporting analytical validation trials.