
Overture Life gains CE mark for automated IVF system
Overture’s DaVitri system aims to make egg thawing and freezing processes during IVF more consistent to improve successful embryo formation outcomes.

Overture’s DaVitri system aims to make egg thawing and freezing processes during IVF more consistent to improve successful embryo formation outcomes.
QuantalX’s Delphi-MD is applicable for the diagnosis, monitoring, and treatment evaluation of a range of neurological conditions.
Paradromics announced today that it received FDA investigational device exemption (IDE) to begin a study of its brain-computer interface (BCI).
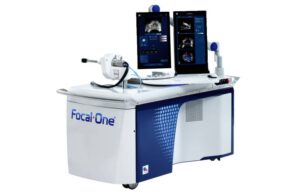
EDAP TMS SA (Nasdaq:EDAP) announced today that the FDA granted 510(k) clearance for new workflows for its ultrasound technology.
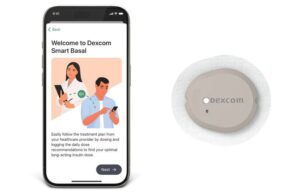
Dexcom (NSDQ:DXCM) announced today that the FDA cleared its Smart Basal CGM-integrated basal insulin dosing optimizer.

ELLSWORTH, Maine, Nov. 19, 2025 /PRNewswire/ — Rejuva Fresh®, a global leader in non-invasive aesthetic and wellness solutions, proudly announces expanded CE certification and design upgrades for its revolutionary EMSTRONG® platform, reinforcing its leadership in pelvic health and intimate wellness.

Having been commercially available for less than 12 months, the system is now offered in over 60 US clinics.

PLANO, Texas, Nov. 18, 2025 /PRNewswire/ — Vesalio, a leader in innovative thrombectomy solutions, today announced two new FDA 510(k) clearances for its aspiration devices, designed for peripheral and neurovascular applications. These clearances mark a key milestone in Vesalio’s evolution toward providing a complete suite of thrombectomy products across multiple vascular territories.

CAMBRIDGE, Mass., Nov. 18, 2025 /PRNewswire/ — Nanopath, a point-of-care diagnostics company enabling high-quality molecular testing in minutes, today announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device designation for the company’s novel assay for the rapid detection of infection in patients with suspected, or at risk of, complicated urinary tract infections (UTIs).
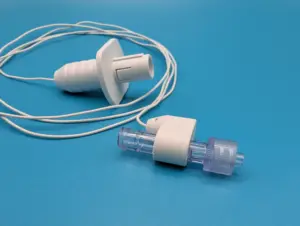
SAN FRANCISCO, Nov. 18, 2025 /PRNewswire/ — Piccolo Medical, Inc. (Piccolo) today announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to expand the Indications for Use for its PM2™ System and ECGuide™ Connector. This clearance extends the technology’s use as an alternative to chest x-ray for a variety of central venous access devices in pediatric and neonatal patient populations.