
Medical Microinstruments picks up reimbursement wins for robotic lymphatic surgery
Medical Microinstruments (MMI) announced today that it won reimbursement for its surgical lymphovenous bypass (LVB) surgery system.

Medical Microinstruments (MMI) announced today that it won reimbursement for its surgical lymphovenous bypass (LVB) surgery system.
The a2z-Unified-Triage device is designed to simultaneously flag and prioritise seven urgent findings on abdomen-pelvis CT scans.

GE HealthCare’s Recon DL is claimed to be the first mammography technology to use deep learning alongside iterative reconstruction to improve image quality.

BD Surgiphor™ is a sterile, ready-to-use irrigation solution designed to help loosen and remove debris from surgical wounds during procedures
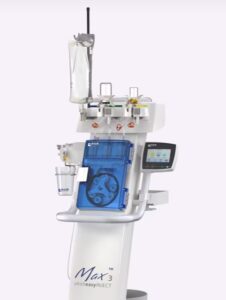
PRINCETON, N.J., Nov. 25, 2025 /PRNewswire/ — Bracco Diagnostics Inc., the U.S. subsidiary of Bracco Imaging S.p.A., a global leader in diagnostic imaging, announced today that the U.S. Food and Drug Administration (FDA) has expanded the indication for the Bracco-branded Max 3™, a Rapid Exchange and Syringeless Injector for use in magnetic resonance imaging (MRI) procedures. In addition to single-dose and multi-dose vials, the Max 3™ system is now indicated for use with the newly approved VUEWAY® (gadopiclenol) injection Imaging Bulk Package (IBP) in 30 mL and 50 mL formats.
Transmural Systems’ Telltale guide wire aims to prevent coronary obstruction by modifying the ‘leaflet’ tissue in high-risk patients undergoing TAVR.

The Centers for Medicare and Medicaid Services (CMS) has provided a major reimbursement update for cardiac ablation technologies.

CeriBell (Nasdaq:CBLL) announced today that it received FDA 510(k) clearance for its next-generation Clarity algorithm.

PARAMUS, N.J., Nov. 25, 2025 /PRNewswire/ — Regenity Biosciences, a global leader in regenerative medicine and Linden Capital Partners portfolio company, today announced its 63rd FDA 510(k) clearance for DuraMatrix® Repair – Collagen Dura Regeneration Membrane, a resorbable membrane composed of highly purified Type I and Type III bovine collagen for the repair of dura mater defects during neurosurgery.

Recor Medical said today that it secured Medicare approval, granting coverage for the company’s RADIANCE CED study.