
FDA approves Nyxoah’s Genio system for obstructive sleep apnoea
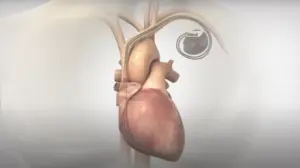
The system is compatible with 1.5T and 3T magnetic resonance imaging scans and eliminates the need for an implanted battery.

The system is compatible with 1.5T and 3T magnetic resonance imaging scans and eliminates the need for an implanted battery.
Orchestra BioMed (Nasdaq:OBIO) announced today that it began the rollout of a protocol update for its BACKBEAT study.

Nyxoah (Nasdaq:NYXH) announced today that it received FDA approval for its Genio neuromodulation device for treating sleep apnea.

SurgiBox, a medical technology company committed to improving access to safe, clean surgery at the point of need, announced today it has received the CE Mark under MDR for the medical devices that comprise its flagship product, the SurgiField System.

BEDFORD, Mass., Aug. 7, 2025 /PRNewswire/ — Instylla, Inc., a privately held company developing novel resorbable embolics for peripheral vascular embolization, announced premarket approval (PMA) from the U.S. Food and Drug Administration (FDA) for the company’s flagship product Embrace™ Hydrogel Embolic System. Embrace HES has been approved for the embolization of hypervascular tumors in peripheral arteries ≤ 5mm.
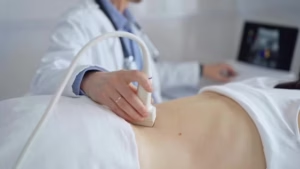
The diagnostic performance of HyCoSy with SF6 microbubbles claims to have been validated by a meta-analysis of 24 trials.
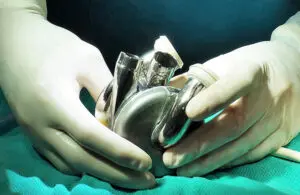
BiVacor announced today that the FDA accepted its Total Artificial Heart (TAH) into its Total Product LifeCycle Advisory Program (TAP).

AbbVie has announced that it has received marketing authorisation from the UK Medicines and Healthcare products Regulatory Agency (MHRA) for ELAHERE (mirvetuximab soravtansine) as monotherapy for the treatment of eligible adult patients with folate receptor-alpha (FRα) positive, platinum-resistant high-grade serous epithelial ovarian, fallopian tube, or primary peritoneal cancer who have received one to three prior systemic treatment regimens.
The RS001 chest-worn device directly measures respiration and provides passive cardio-respiratory monitoring.
Regulatory green light marks full commercial launch of LungVision, enhancing early lung cancer detection through AI-driven 3D imaging for more precise bronchoscopic diagnostics.