FDA grants breakthrough device designation for Quest Diagnostics’ MRD test
The liquid biopsy test has been developed by experts in cancer genomics.

The liquid biopsy test has been developed by experts in cancer genomics.
Brightonix Imaging announced today that its flagship Pharos positron emission tomography (PET) scanner received FDA clearance.

The stent is engineered to keep arteries open, deliver everolimus for vessel healing, and gradually dissolve over time.

Abbott (NYSE: ABT)+
announced today that it received CE mark for its Esprit BTK everolimus-eluting resorbable scaffold system.
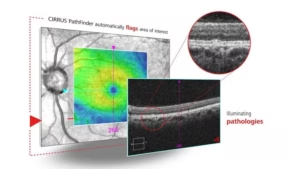
The CIRRUS PathFinder tool is available via licensing in the new software update.
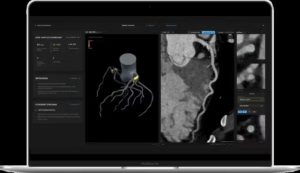
PERTH, Australia, Aug. 21, 2025 /PRNewswire/ — Artrya Limited (ASX: AYA) (Artrya or the Company), a medical technology company commercialising its Salix® AI-powered cloud platform, for the near real time, point of care assessment and management of coronary artery disease, is pleased to announce it has received 510(k) clearance from the U.S. Food and Drug Administration (the FDA) for Artrya’s proprietary, Salix® Coronary Plaque module.

Cepheid announced that the test will be shipped to Canadian customers this month.
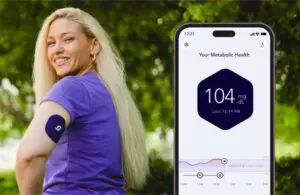
Signos today announced a major step forward in bringing over-the-counter (OTC) continuous glucose monitoring (CGM) to market.

Microbot Medical (Nasdaq:MBOT) announced today that it received a new patent covering a modular robotic surgical system.
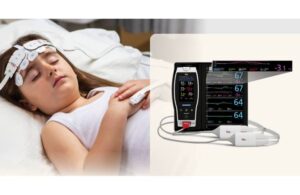
Masimo (Nasdaq: MASI)+
announced today that it received FDA 510(k) clearance for expanded indications for its O3 regional oximetry technology