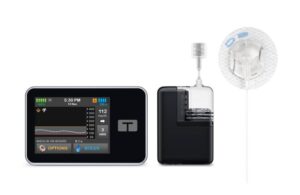
Tandem picks up FDA clearance for 7-day infusion set
Tandem Diabetes Care (Nasdaq:TNDM) has a new infusion set on the way to pair with its insulin delivery offerings.

Tandem Diabetes Care (Nasdaq:TNDM) has a new infusion set on the way to pair with its insulin delivery offerings.

ANKARA, Türkiye, Aug. 18, 2025 /PRNewswire/ — Nerveblox, an AI software solution by SmartAlpha, designed to assist physicians in using ultrasound while administering regional anesthesia procedures commonly known as ‘nerve blocks’, has received U.S. Food and Drug Administration (FDA) 510(k) clearance.
The company is collaborating with local companies and healthcare providers to distribute the ulcer test.
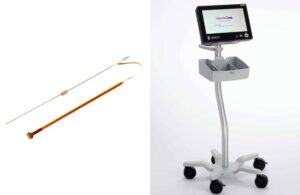
NeuroOne (Nasdaq:NMTC) announced today that it received FDA clearance for its OneRF trigeminal nerve ablation system.

Onward Medical announced today that it received FDA investigational device exemption (IDE) for its ARC-IM system.

The test was previously granted a breakthrough device designation by the US regulator.

DeepSight Technology announced today that it received FDA 510(k) clearance for its NeedleVue LC1 ultrasound system
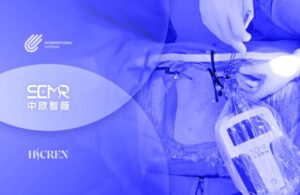
Interventional Systems and HICREN today announced approval for their joint venture’s surgical robot platform in China.
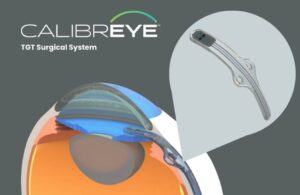
Myra Vision announced today that it received FDA investigational device exemption (IDE) approval for its Calibreye system
Renata Medical announced today that CMS granted a New Technology Add-on Payment (NTAP) for its Minima stent system.