GE HealthCare wins FDA clearance for new coronary CT system
GE HealthCare (Nasdaq: GEHC)+
announced that it received FDA 510(k) clearance for its Revolution Vibe CT imaging system.

GE HealthCare (Nasdaq: GEHC)+
announced that it received FDA 510(k) clearance for its Revolution Vibe CT imaging system.

Surgerii Robotics announced on LinkedIn today that its Shurui single-port surgical robot garnered CE mark approval in Europe.
The submission for CT:VQ was supported by a clinical validation package that spanned a variety of lung conditions.

Imperative Care announced today that the FDA granted 510(k) clearance for its Symphony thrombectomy system to treat pulmonary embolism (PE).

Medtronic (NYSE: MDT)+
today announced major regulatory milestones for its MiniMed 780G automated insulin delivery system.

Abbott (NYSE: ABT)+
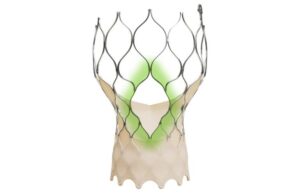
announced today that it received CE mark for an expanded indication for its Navitor TAVI system.
Medtronic (NYSE: MDT)+
announced today that it received FDA approval for the expanded redo-TAVR indication of its Evolut system.

Cartherics, a biotech company developing off-the-shelf immune cell therapies focusing on high-impact women’s diseases, with lead programs in ovarian cancer and endometriosis, has announced the granting of a patent for ‘Method for Providing Immune Cells with Enhanced Function’ by the Chinese Patent Office.

Ciliatech announced today that it received CE mark approval for its Intercil uveal spacer, an eye implant for treating glaucoma.
Airiver Medical announced today that it received FDA investigational device exemption (IDE) for its pulmonary drug-coated balloon (DCB).