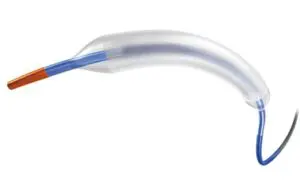
FDA approves Boston Scientific’s Agent drug-coated balloon
Boston Scientific (NYSE: BSX)+
announced today that it received FDA approval for its Agent drug-coated balloon (DCB).

Boston Scientific (NYSE: BSX)+
announced today that it received FDA approval for its Agent drug-coated balloon (DCB).
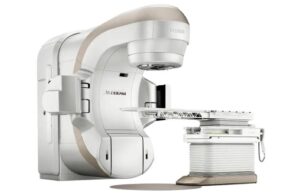
Varian announced today that it received FDA 510(k) clearance for its TrueBeam and Edge radiotherapy systems with HyperSight imaging.

FDA Clears Soldier Microcatheter for Localized Drug Delivery in Vascular Interventions
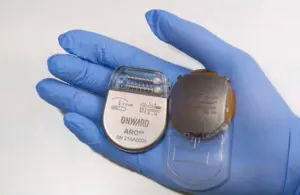
Onward Medical announced today that the FDA granted breakthrough device designation for its ARC-BCI system that uses brain-computer interface (BCI) technology.

TYLER, Texas–(BUSINESS WIRE)– AIOMEGA, a Texas biomedical company, announced that AIO BREATHE, their medical device that treats Obstructive Sleep Apnea, has been cleared by the Food and Drug Administration.

Nevro1™ Proven to Immediately Transfix Sacroiliac (SI) Joint to Allow for Long-term SI Joint Fusion
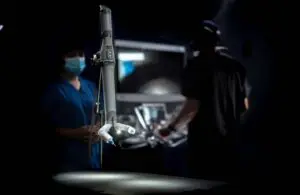
Virtual Incision announced that the FDA granted marketing authorization to its MIRA miniaturized surgical robotic system.

The clearance poises the orthopedics company to be first to market with robotic-assisted shoulder replacement surgery.

For use in children weighing 10 kilograms or more with acute kidney injury (AKI) due to sepsis or a septic condition requiring kidney replacement therapy (KRT).
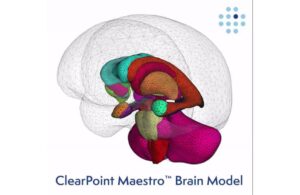
ClearPoint Neuro (Nasdaq:CLPT) announced that the FDA cleared its ClearPoint 2.2 software with integrated Maestro Brain Modeling.