
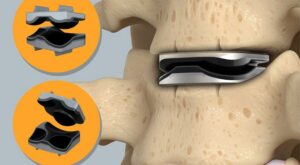
SurGenTec gains new FDA indication for ION-C facet fixation system
SurGenTec’s ION-C system is now indicated for the treatment of cervical pseudoarthrosis when implanted bilaterally within the facet joints.

SurGenTec’s ION-C system is now indicated for the treatment of cervical pseudoarthrosis when implanted bilaterally within the facet joints.
MORGANTOWN, W.Va., Jan. 20, 2026 /PRNewswire/ — Omnia Medical, a medical technology company developing surgical solutions for spine and interventional pain physicians, today announced the commercial launch of its FDA-cleared PsiF DNA™ Sacroiliac Joint Stabilization System.
SEATTLE, Jan. 15, 2026 /PRNewswire/ — Proprio, the surgical technology company pioneering real-time, AI-powered intraoperative guidance and data-driven surgical workflows, today announced the U.S. Food and Drug Administration (FDA) has granted clearance for the “Picasso” feature. This marks the company’s fourth FDA-cleared capability within its Paradigm platform.
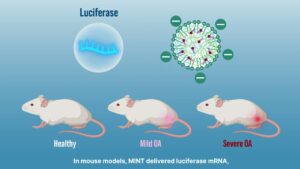
Nanoparticles are engineered to home in on areas where cartilage has degenerated in osteoarthritis, ensuring that treatment concentrates exactly where it is needed
When a house catches on fire, we assume that a smoke alarm inside will serve one purpose and one purpose only: warn the occupants of danger. But imagine if the device could transform into something that could fight the fire as well.

A comprehensive genetic investigation led by Dr. Feng Liu at Tianjin Medical University General Hospital has uncovered striking molecular connections between schizophrenia and bone health, identifying 195 shared genetic loci that may explain why psychiatric patients face elevated fracture risks.

Cedars-Sinai investigators have discovered a healing mechanism that could one day be harnessed to help treat patients with spinal cord injuries, stroke, and neurological conditions such as multiple sclerosis. Their study, published in Nature, describes a previously unknown function of astrocytes, a type of cell in the central nervous system.
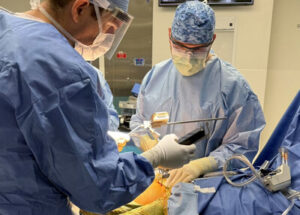
IRVINE, Calif., Dec. 16, 2025 /PRNewswire/ — OrthAlign, Inc., a global leader in surgical navigation technologies, today announced the expansion of its Lantern® Hip platform to include support for posterior-based approaches in total hip arthroplasty (THA).

TAMPERE, Finland, Dec. 14, 2025 /PRNewswire/ — Bioretec Oy (“Bioretec” or the “Company”), a pioneer in biodegradable orthopedic implants, has been granted Breakthrough Device Designation by the U.S. Food and Drug Administration (FDA) for its magnesium alloy technology-based, biodegradable RemeOs™ DrillPin.

Partnership will expand Shoulder Innovations’ Disruptive Ecosystem with Advanced Enabling Technology, Complementing Surgeon and Patient Needs in the ASC