New ultra-small coil enables precise brain stimulation
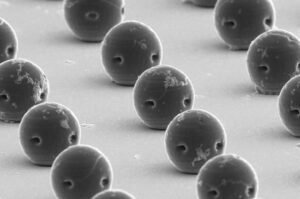
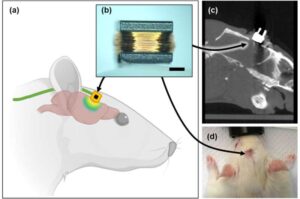
A research team led by Prof. Kim So-hee from the Department of Robotics and Mechanical Electronics, DGIST, has developed a technology that enables precise brain stimulation using a coil small enough to be implanted in the body. It is expected to be utilized as an electronic medicine for brain neurological disorders that require long-term treatment due to its ability to significantly improve safety and effectiveness with fewer side effects compared to existing technologies.