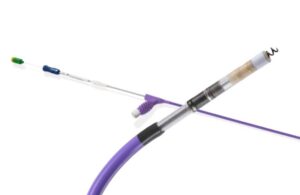
FDA approves expanded label for Boston Scientific Ingevity+ pacing leads
Boston Scientific (NYSE: BSX)+
announced today that it received FDA approval to expand the indication for its current-generation Ingevity+ pacing leads.

Boston Scientific (NYSE: BSX)+
announced today that it received FDA approval to expand the indication for its current-generation Ingevity+ pacing leads.
Philips (NYSE: PHG)+
announced today that it introduced the FDA-approved, 160 cm version of its LumiGuide endovascular navigation wire.

Edwards Lifesciences (NYSE: EW)+
announced that it launched its Sapien 3 transcatheter pulmonary valve implant (TPVI) system with Alterra adaptive prestent in Europe.

Medtronic (NYSE: MDT)+
today announced the publication of its novel approach to studying adaptive deep brain stimulation (aDBS).

Commercial partner Ascensia is in discussions with insulin pump manufacturers to create an automated insulin delivery system.

Biotronik announced today that it received FDA approval for its Selectra 3D catheter and Solia S lead for use in left bundle branch area pacing (LBBAP).

The company says that Lightning Flash 2.0 will be the most advanced thrombectomy system on the European market to address venous and pulmonary thrombus.

Johnson & Johnson MedTech‘s Shockwave Medical today announced the full U.S. launch of its Shockwave E8 peripheral IVL catheter.
SamanTree Medical announced today that it received FDA 510(k) clearance for its Histolog scanner for tissue imaging.

SILVER SPRING, Md., Sept. 12, 2024 /PRNewswire/ — Today, the U.S. Food and Drug Administration authorized the first over-the-counter (OTC) hearing aid software device, Hearing Aid Feature, intended to be used with compatible versions of the Apple AirPods Pro headphones. Once installed and customized to the user’s hearing needs, the Hearing Aid Feature enables compatible versions of the AirPods Pro to serve as an OTC hearing aid, intended to amplify sounds for individuals 18 years or older with perceived mild to moderate hearing impairment.