
From 3 days to 45 minutes: rapid test for UTI wins $10m Longitude Prize on AMR to transform fight against superbugs
Challenge Works today announces that Sysmex Astrego’s PA-100 AST System has won the $10m (£8m) Longitude Prize on AMR.

Challenge Works today announces that Sysmex Astrego’s PA-100 AST System has won the $10m (£8m) Longitude Prize on AMR.
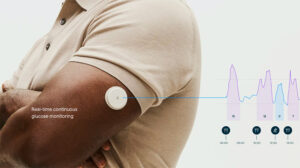
Abbott (NYSE: ABT)+ today announced FDA clearance for two over-the-counter continuous glucose monitor (CGM) systems, Lingo and Libre Rio.
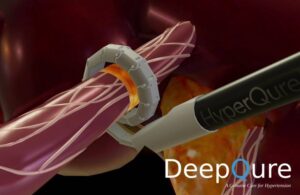
DeepQure announced today that it received FDA investigational device exemption (IDE) approval for a study of its HyperQure system.

CroíValve today announced favorable patient outcomes in a first-in-human clinical trial of its Duo tricuspid coaptation valve system.

Johnson & Johnson MedTech‘s DePuy Synthes announced today that it received a new FDA 510(k) clearance for its Velys surgical robot platform.

Mequ announced today that the FDA granted 510(k) clearance to its M Warmer system, a portable blood and IV fluid warmer platform.
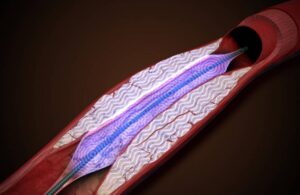
Amplitude Vascular Systems announced today that it received FDA investigational device exemption (IDE) for its pulsatile intravascular lithotripsy (PIVL) therapy.

Abbott (NYSE: ABT)+ announced today that it received CE mark approval for its Aveir dual-chamber (DR) leadless pacemaker system.
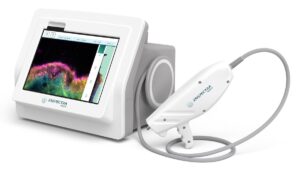
Enspectra Health, a health tech company, today announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation for its next generation AI-powered VIO™ Skin Platform (VIO)

FloPatch from Flosonics Medical provides real-time patient data to enhance clinical decision-making during life-saving critical care events