
Profound Medical wins FDA clearance for AI to support prostate cancer procedure
Profound Medical (Nasdaq:PROF) announced today that it received FDA 510(k) clearance for its second AI model for treating prostate cancer.

Profound Medical (Nasdaq:PROF) announced today that it received FDA 510(k) clearance for its second AI model for treating prostate cancer.

The FDA has warned that home test users, caregivers and healthcare providers should not use Cue Health COVID-19 diagnostics.

Carthera announced today that it received FDA and European orphan drug designation (ODD) for the use of carboplatin with its SonoCloud.

CENTER VALLEY, Pa., May 13, 2024 /PRNewswire/ — Olympus, a global medical technology company, announced today the launch of two bronchoscopes as part of the EVIS X1 Endoscopy System. The EVIS X1 Endoscopy System represents the latest in diagnostic and therapeutic bronchoscopy from Olympus.

Medtronic (NYSE: MDT)+
announced today that its PulseSelect pulsed field ablation (PFA) system has won regulatory approval in Japan.

LAKEWOOD, Colo., May 9, 2024 /PRNewswire/ — Terumo Blood and Cell Technologies (Terumo BCT), a medical technology company, recently received U.S. Food and Drug Administration (FDA) 510(k) clearance for the Rika Plasma Donation System™ with the iNomi™ Nomogram. This innovation means that plasma collection volume can be determined by an individual donor’s height, weight and hematocrit level on the day they donate plasma.
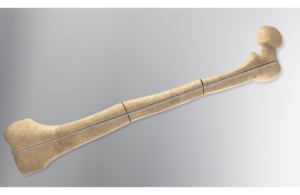
Orthofix (Nasdaq: OFIX)+
today announced it received FDA 510(k) clearance for its Rodeo Telescopic Nail.

OrthoXel announced that it received FDA 510(k) clearance for its Vertex hip fracture nail (HFN) for fracture fixation.
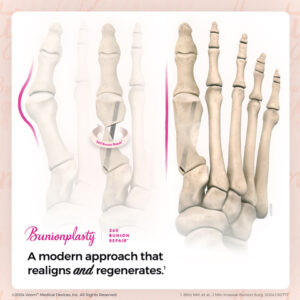
The company is pleased to report a rapidly accelerating interest in its proprietary, minimally invasive Bunionplasty® 360 Bunion Repair™ solution
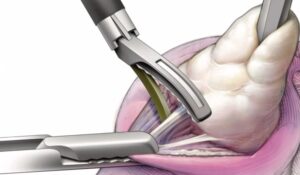
Freyja Healthcare Brings First-of-Its-Kind 2mm Abdominal-Access Device to Laparoscopic Surgery to Further Innovation in Fast-Growing Women’s Health Market