Amid US ordeal, Moderna wins EU approval for flu/COVID-19 combo shot
In the U.S., Moderna withdrew its approval application for the combination vaccine in May last year and the timeline for resubmission remains uncertain.

In the U.S., Moderna withdrew its approval application for the combination vaccine in May last year and the timeline for resubmission remains uncertain.
The ArteraAI prostate biopsy assay predicts the risk of prostate cancer progression.
Approval for Xeltis’s aXess device was based on pivotal trial data that demonstrated 79% secondary patency and a patency related reintervention rate of 1.3 per patient year.
Johnson & Johnson (NYSE:JNJ) announced today that it received CE mark approval for its new Ethicon 4000 surgical stapler.
Philips (NYSE:PHG) announced today that it received FDA 510(k) clearance for its Rembra platform of scanning systems.
Sonorous Neurovascular announced today that it received FDA 510(k) clearance for its BosCATH next-generation neurovascular catheter.

Luminoah announced today that it received FDA clearance for Luminoah Flow, an enteral feeding system for those who rely on tube feeding.
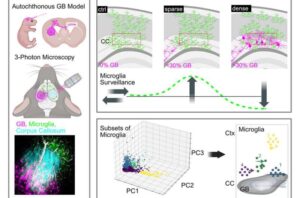
Researchers from DZNE, University Hospital Bonn and the Cluster of Excellence ImmunoSensation at the University of Bonn have captured this infiltration process in the living brain with advanced microscopy.
Oregon Health & Science University researchers have developed a first-of-its-kind method to predict cancer patient survival using advanced molecular data from individual cells.
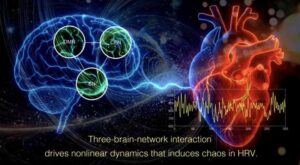
A team of researchers at Kyoto University have demonstrated that the chaotic component of heartbeat variability is uniquely sensitive to cognitive brain activity. Conventional heart rate variability, HRV, indices show no consistent response, whereas chaos-based measures reveal clear and reproducible changes, providing a new noninvasive indicator of brain-heart interaction.