
Endo Tools Therapeutics Receives FDA 510(k) Clearance for Next-Generation endomina® EZFuse system
510(k) clearance enables immediate U.S. commercialization of enhanced soft tissue approximation technology in the gastrointestinal tract

510(k) clearance enables immediate U.S. commercialization of enhanced soft tissue approximation technology in the gastrointestinal tract

CHICAGO, March 17, 2026 /PRNewswire/ — Introducing DERMAFLASH® COOL, a game-changing innovation from the industry leader in sonic skincare technology. Unlike cryotherapy tools that require freezing, this patented device is powered by rechargeable SonicCool™ + Copper Technology and reaches an industry-leading low temperature (the coldest on the market) in seconds.
An innovative platform developed by PKU researchers called “cf-EpiTracing” has proved capable of detecting and tracing diseases from as little as 50 μl of human plasma, or roughly a drop of blood. The research, published in Nature on March 4, 2026, was led by Professor He Aibin from the College of Future Technology and Professor Jing Hongmei from the Department of Hematology, PKU Third Hospital.
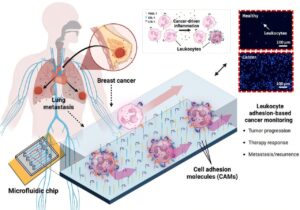
A new microfluidic technology that leverages immune cell behavior is set to transform cancer monitoring, thanks to researchers at UNIST. Led by Professor Joo Hun Kang in the Department of Biomedical Engineering at UNIST, the team has introduced a novel diagnostic chip that analyzes the adhesion properties of leukocytes, or white blood cells, to detect cancer recurrence and evaluate the effectiveness of chemotherapy.

“The smart mask is a low-cost way of providing continuous health care monitoring.”

The technology could enable fast, point-of-care diagnoses for pneumonia and other lung conditions.
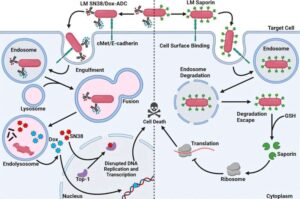
Baylor University researchers have developed a novel approach to fight colorectal cancer, using modified bacteria as a courier to deliver potent cancer-killing proteins into tumor cells.

From cobblers to smoothies, dark sweet cherries show up in plenty of recipes, and scientists say the crimson-colored fruit may contain compounds that could help fight an aggressive type of breast cancer.
According to multiple reports, the Chinese medical device regulatory body granted Neuracle the country’s first approval for a brain-computer interface (BCI).
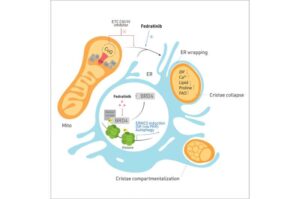
Cells behave like cities and organelles carry out infrastructural roles: mitochondria are powerhouses, the endoplasmic reticulum serves as a transport hub and lysosomes help with waste disposal.