MedTech News
.................... by Andrew Celentano
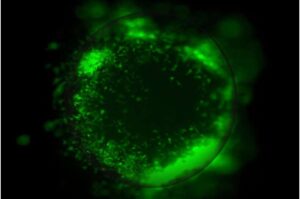
Lab-grown organoids reveal how glioblastoma outsmarts treatment
UCLA scientists have developed advanced miniature 3D tumor organoid models that make it possible to study glioblastoma tumors in a setting that closely mirrors the human brain, shedding light on how the aggressive cancer interacts with surrounding brain cells and the immune system to become more invasive and resistant to therapy.
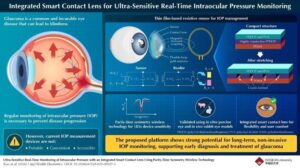
Smart contact lens with thin-film sensor enables real-time eye pressure monitoring
Glaucoma is a leading cause of blindness among people who are unable to monitor and manage their intraocular pressure (IOP) daily. The current tools for IOP measurement are not portable, convenient, easily accessible, or capable of continuous (24/7) monitoring.

New study finds heart attacks involve brain and immune system, not just heart
The conventional approach to studying and treating these episodes is to focus on the heart as an isolated organ. University of California San Diego research, led by the School of Biological Sciences, is upending the way heart attacks are viewed under a transformative new understanding of how cardiac events are interconnected with other systems.

Scientists develop first gene-editing treatment for skin conditions
Gene-editing tools like CRISPR have unlocked new treatments for previously uncurable diseases. Now, researchers at the University of British Columbia are extending those possibilities to the skin for the first time.
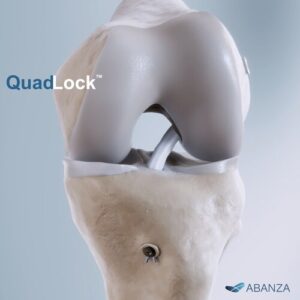
ABANZA Secures FDA 510(k) Clearance for QuadLock™ Fixation System for ACL Reconstruction
ORLANDO, Fla., Jan. 27, 2026 /PRNewswire/ — ABANZA, a company focused on next-generation soft-tissue repair solutions, today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for QuadLock™, its new knotless, bidirectional tension-adjustable fixation system for ACL reconstruction.
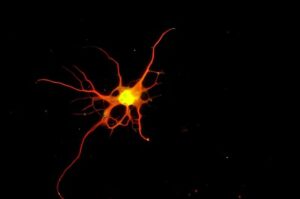
Scientists grow specialized nerve cells that degenerate in ALS and are damaged in spinal cord injury
Researchers have developed a way to grow a highly specialized subset of brain nerve cells that are involved in motor neuron disease and damaged in spinal injuries
Leftover COVID spike fragments kill crucial immune cells but are less deadly in omicron
New research shows that after the body’s defenses kill the virus behind COVID-19, leftover digested chunks of SARS-CoV-2 spike protein can target specific immune cells based on their shape.

Covestro unveils Maestro device for improved diagnostic testing
“The Maestro combines advanced functionality with more sustainable design and electronic integration, making it ideal for today’s clinical and home healthcare applications,” said Eric Saks.
